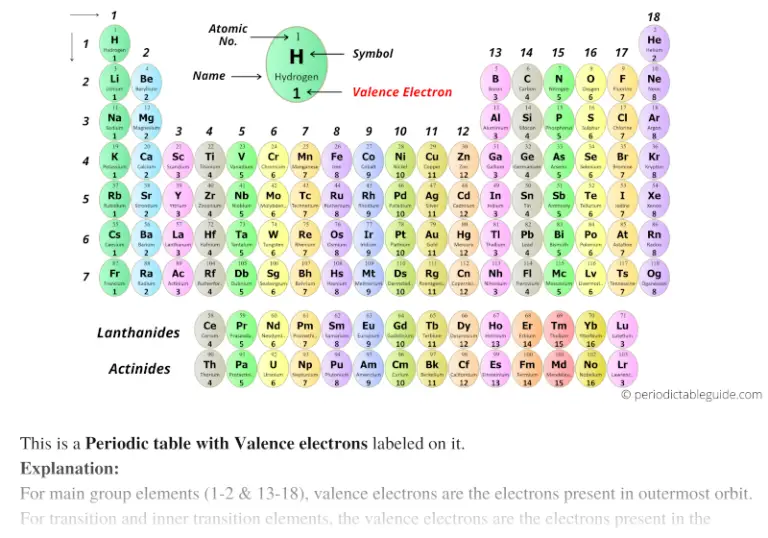
Each hydrogen atom has one valence electron and is univalent. Carbon has four valence electrons and here a valence of four. The only reasonable Lewis electron dot diagram for this compound has the P atom making five covalent bonds:įormally, the P atom has 10 electrons in its valence shell.An electron in the outer shell of an atom's energy levels Four covalent bonds. Such compounds are formed only by central atoms in the third row of the periodic table or beyond that have empty d orbitals in their valence shells that can participate in covalent bonding. These are called expanded valence shell molecules. The third violation to the octet rule is found in those compounds with more than eight electrons assigned to their valence shell. For example, beryllium can form two covalent bonds, resulting in only four electrons in its valence shell:īoron commonly makes only three covalent bonds, resulting in only six valence electrons around the B atom. The most common examples are the covalent compounds of beryllium and boron. These stable compounds have less than eight electrons around an atom in the molecule. Although NO is a stable compound, it is very chemically reactive, as are most other odd-electron compounds.Įlectron-deficient molecules represent the second violation to the octet rule. The Lewis electron dot diagram for NO is as follows:Īlthough the O atom has an octet of electrons, the N atom has only seven electrons in its valence shell. Examples of stable odd-electron molecules are NO, NO 2, and ClO 2. With an odd number of electrons, at least one atom in the molecule will have to violate the octet rule. Although they are few, some stable compounds have an odd number of electrons in their valence shells. Odd-electron molecules represent the first violation to the octet rule. There are three violations to the octet rule. As with many rules, there are exceptions, or violations. This does not mean that the octet rule is useless-quite the contrary. Always make sure all valence electrons are accounted for and each atom has an octet of electrons except for hydrogen (with two electrons).Īs important and useful as the octet rule is in chemical bonding, there are some well-known violations.This will not change the number of electrons on the terminal atoms.If the central atom has fewer electrons than an octet, use lone pairs from terminal atoms to form multiple (double or triple) bonds to the central atom to achieve an octet. We will explain later that some atoms are able to accommodate more than eight electrons.Ħ.If any electrons are left over, place them on the central atom. These electrons will usually be lone pairs.ĥ.Beginning with the terminal atoms, add enough electrons to each atom to give each atom an octet (two for hydrogen). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

:max_bytes(150000):strip_icc()/PeriodicTableCharge-BBG-58b5c80a3df78cdcd8bbb6c8.png)

 RSS Feed
RSS Feed
